Description
The operating software is free! And it will continue to be updated!
Buy more cheap more!
If you want a better price, please send me an offer!
| Comparison Item | Intraoral Scanner V3 (3.0 Pro) | High-Speed Intraoral Scanner V5 |
|---|---|---|
| Handle Weight | 204g | 160g (44g lighter, less hand fatigue) |
| Scanning Depth of Field | 0–15mm | 0–25mm (higher scanning tolerance) |
| Full Arch Scanning Time | 60–90 seconds | 30–50 seconds (higher clinical efficiency) |
| Scanning Accuracy | Single tooth ≤15μm; Full mouth ≤30μm | Same as V3, top-level precision maintained |
| Scan Tips Configuration | 2 standard tips + 1 small tip | 3 standard tips + 1 small tip |
| Housing Design | Spliced structure | Integrated seamless body, easy to disinfect |
| Grip & Operation | Traditional design | Ergonomic optimized, more comfortable grip |
V3







Not include Scanner Cart and Computer !!! 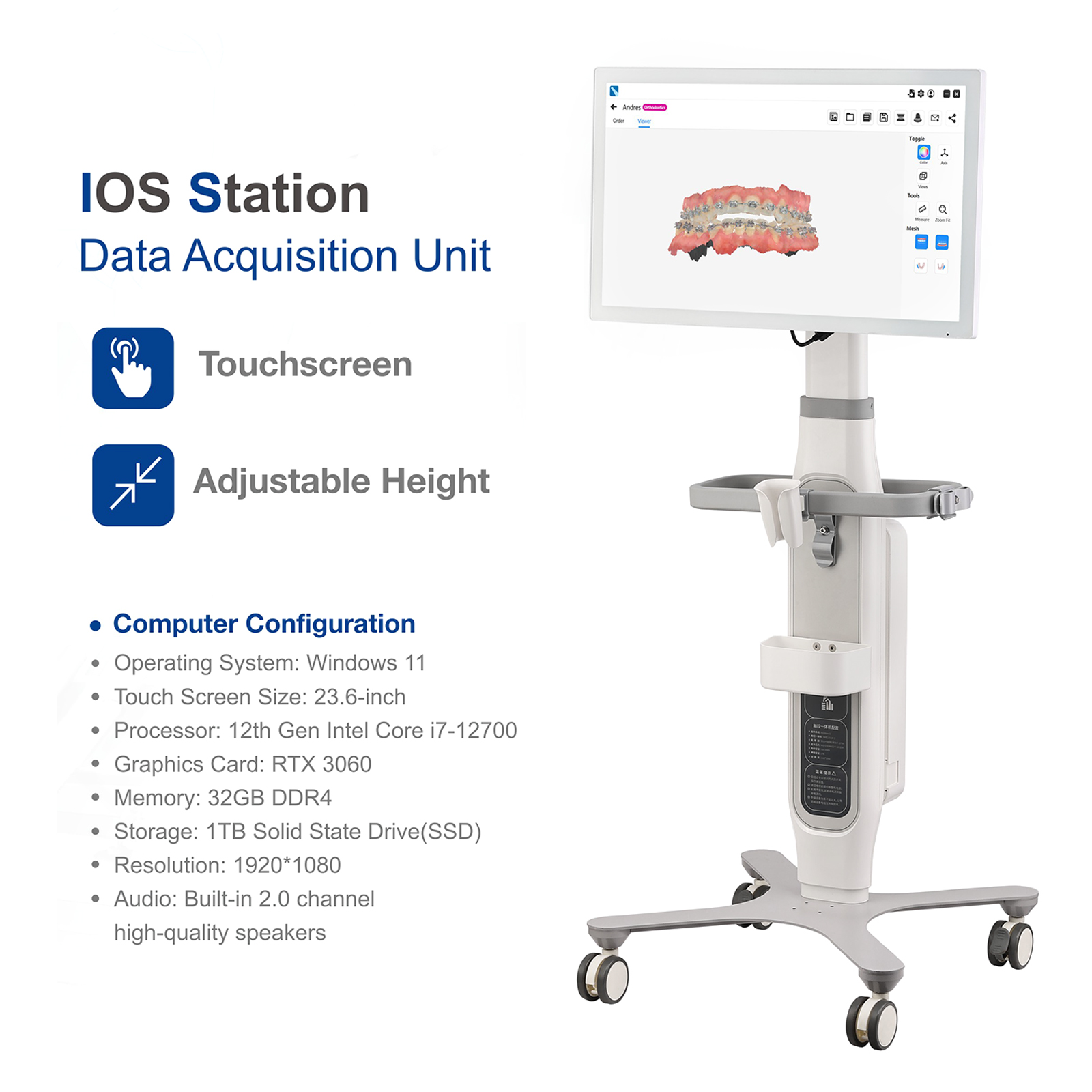

V5

Fast, Accurate, Intelligent
Brand New Intraoral Scanner — 3DS V5
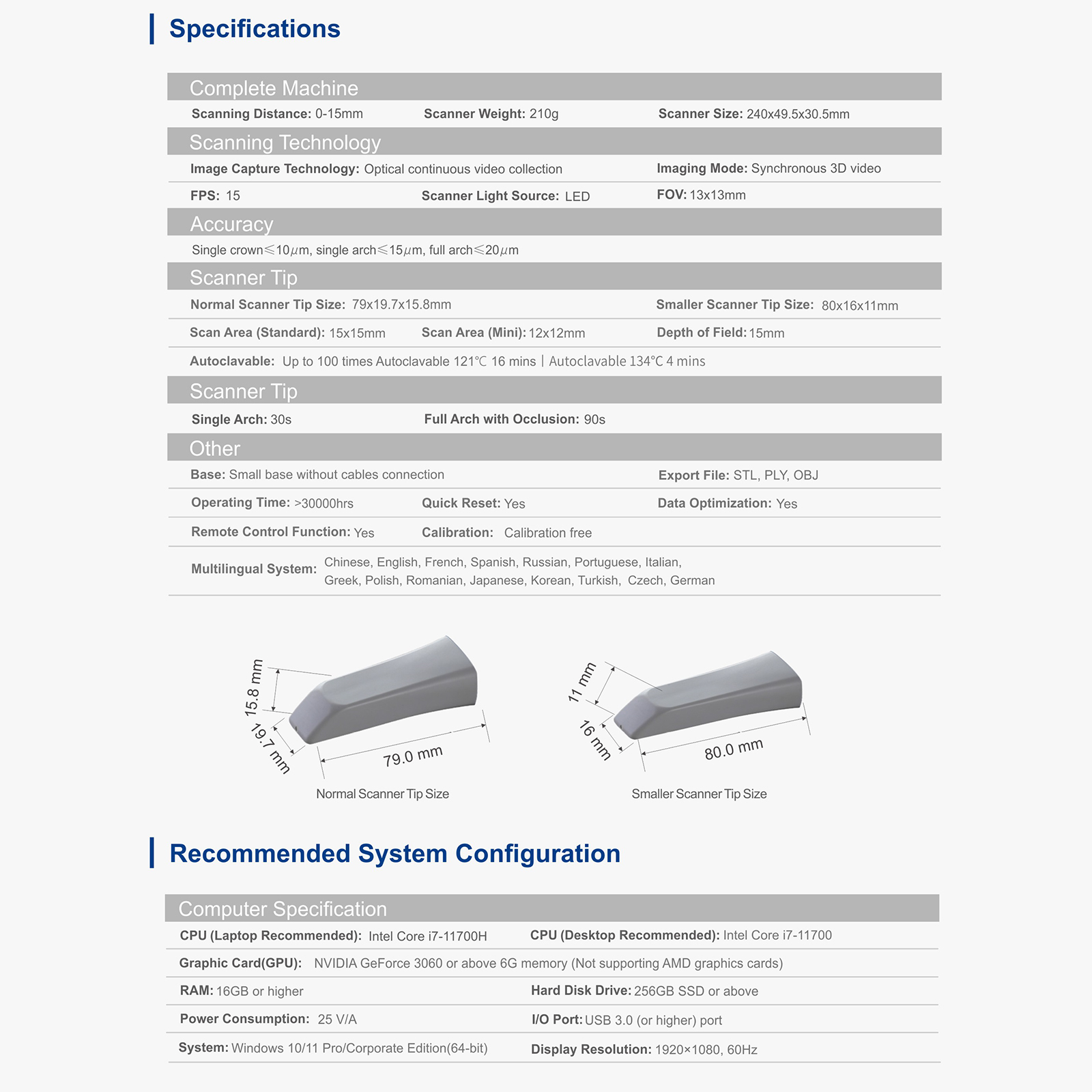
Specifications
- Quick Reset: Yes
- Connection: Wired USB 3.0
- Handpiece Weight: 160g (without cable)
- Depth of View: 0–25mm
- Scanning Accuracy: Single Crown ≤10μm, Full Arch ≤20μm
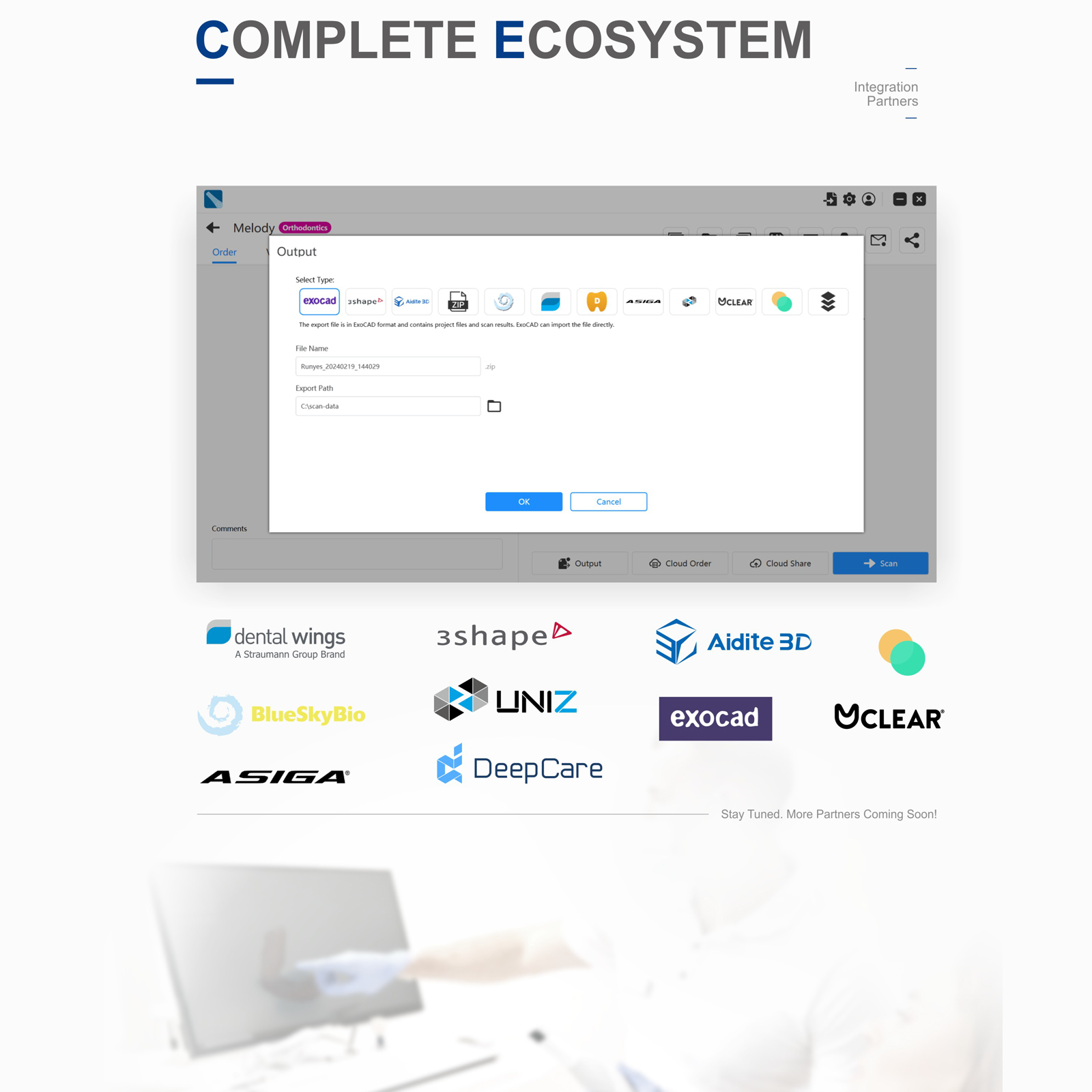
- Output Format: STL, PLY (True Color), OBJ
- Anti-fog: Fan-cooling
- Scanner Tips: 3 Standard + 1 Mini + 1 Protective Cover
- Calibration Tip: 1 Set (Optional)
- Product Lifespan: 8 Years
- Tip Size: Standard 115×21×16mm / Mini 115×17×13mm
- Scanning Area: Standard 16×14mm / Mini 12×10mm
- Imaging: Optical Continuous Video Capture
- Image Mode Synchronization: 3D Video
Product Advantages
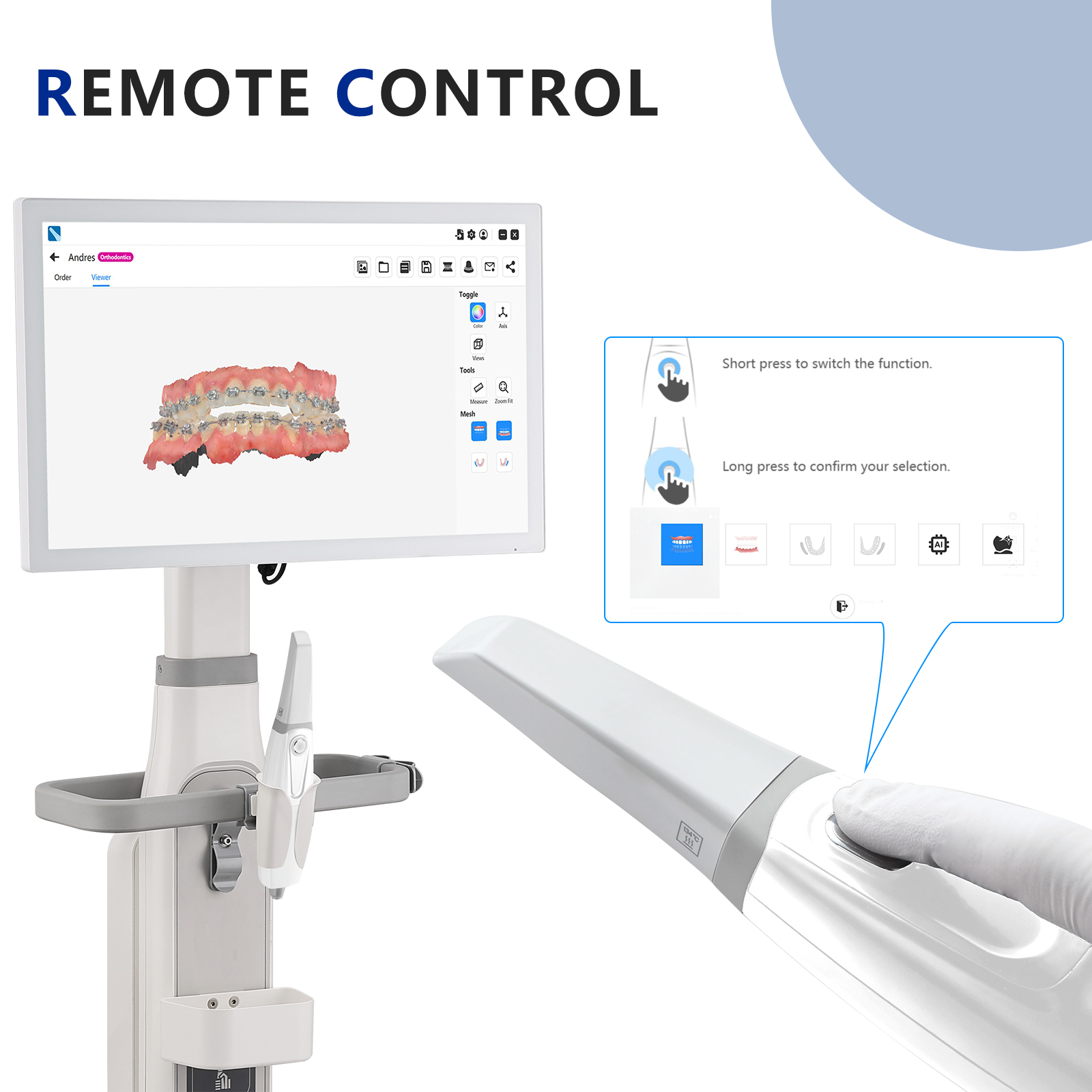
- AI Smart Scanning
- Metal Scanning
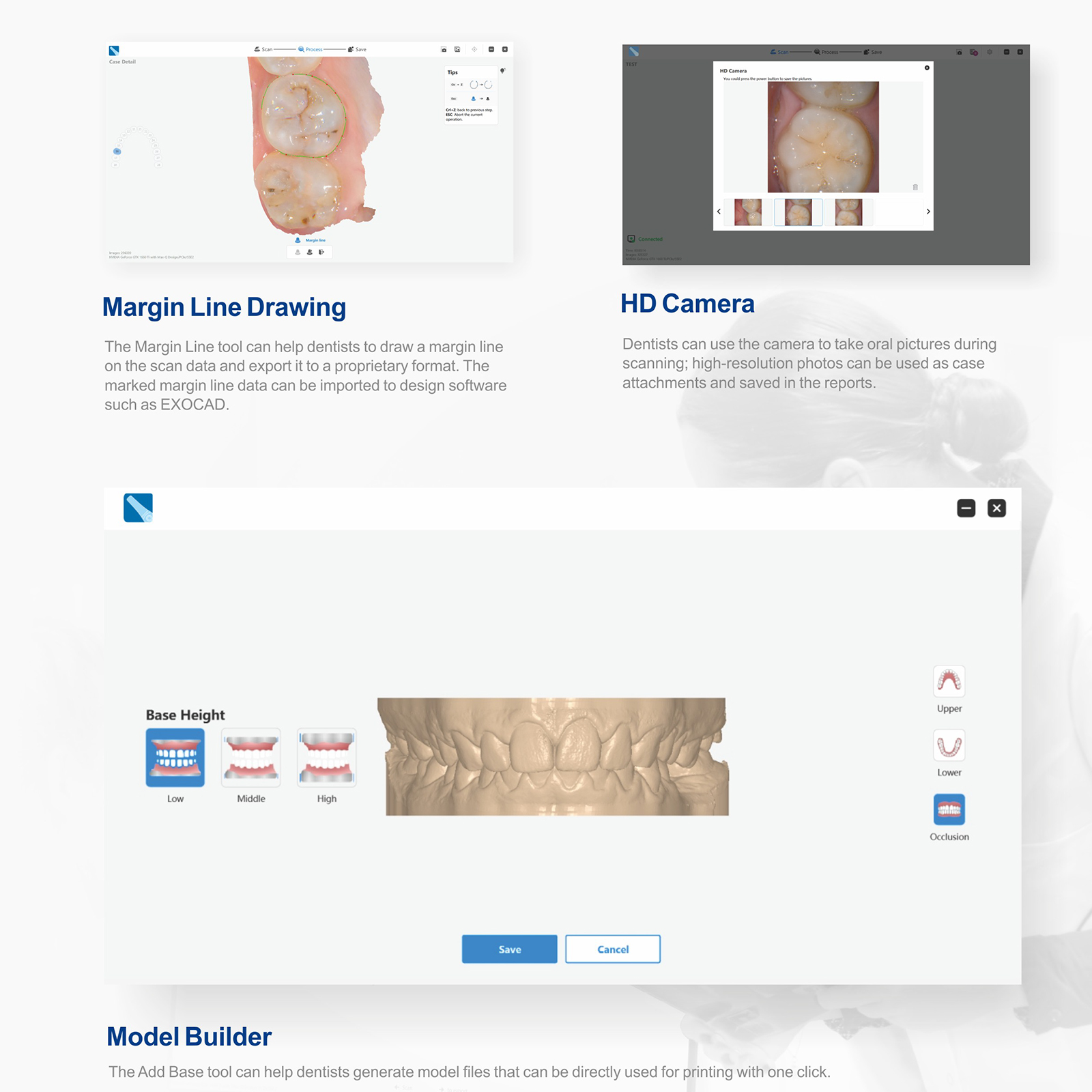
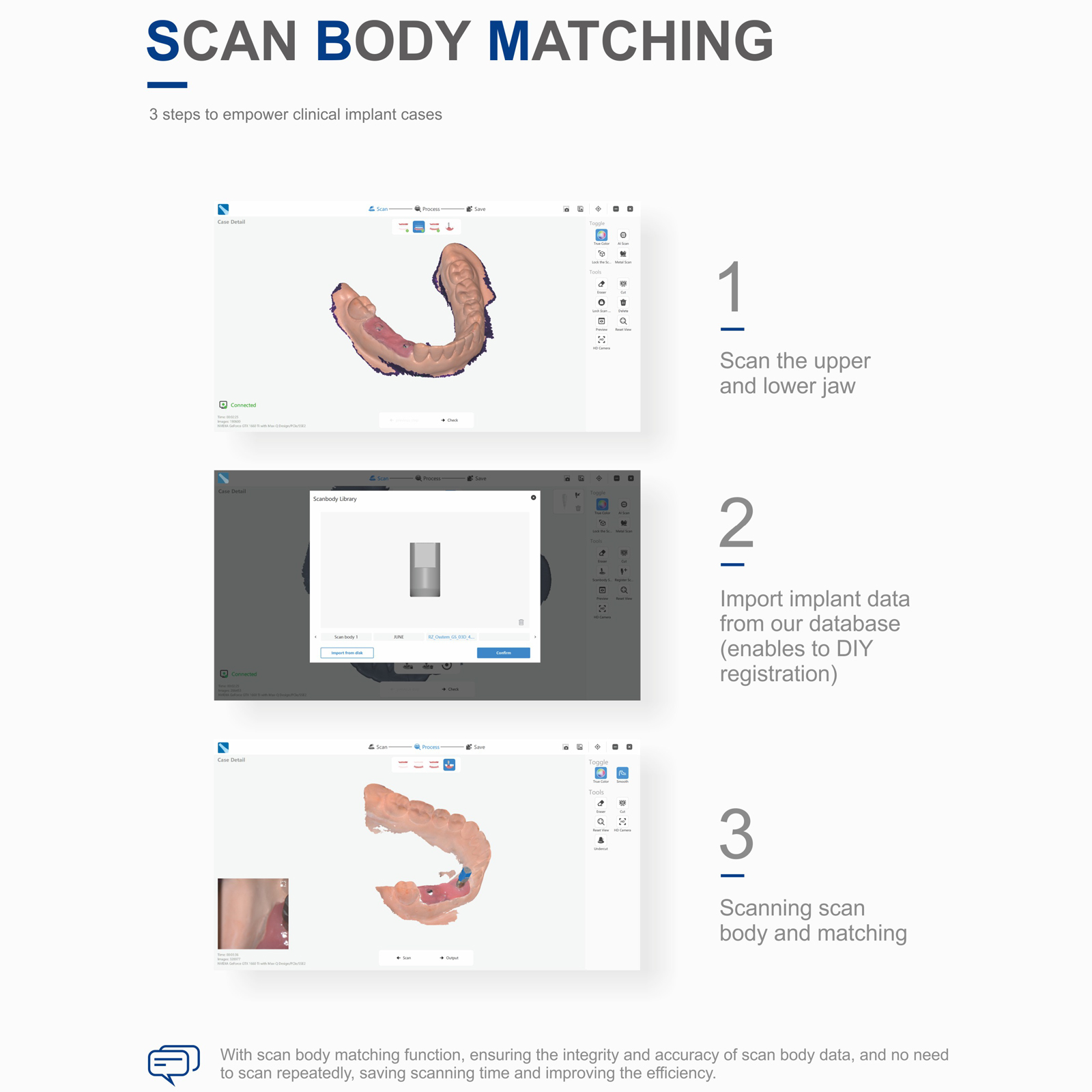
- Scan Body Smart Matching
- Oral Health Report
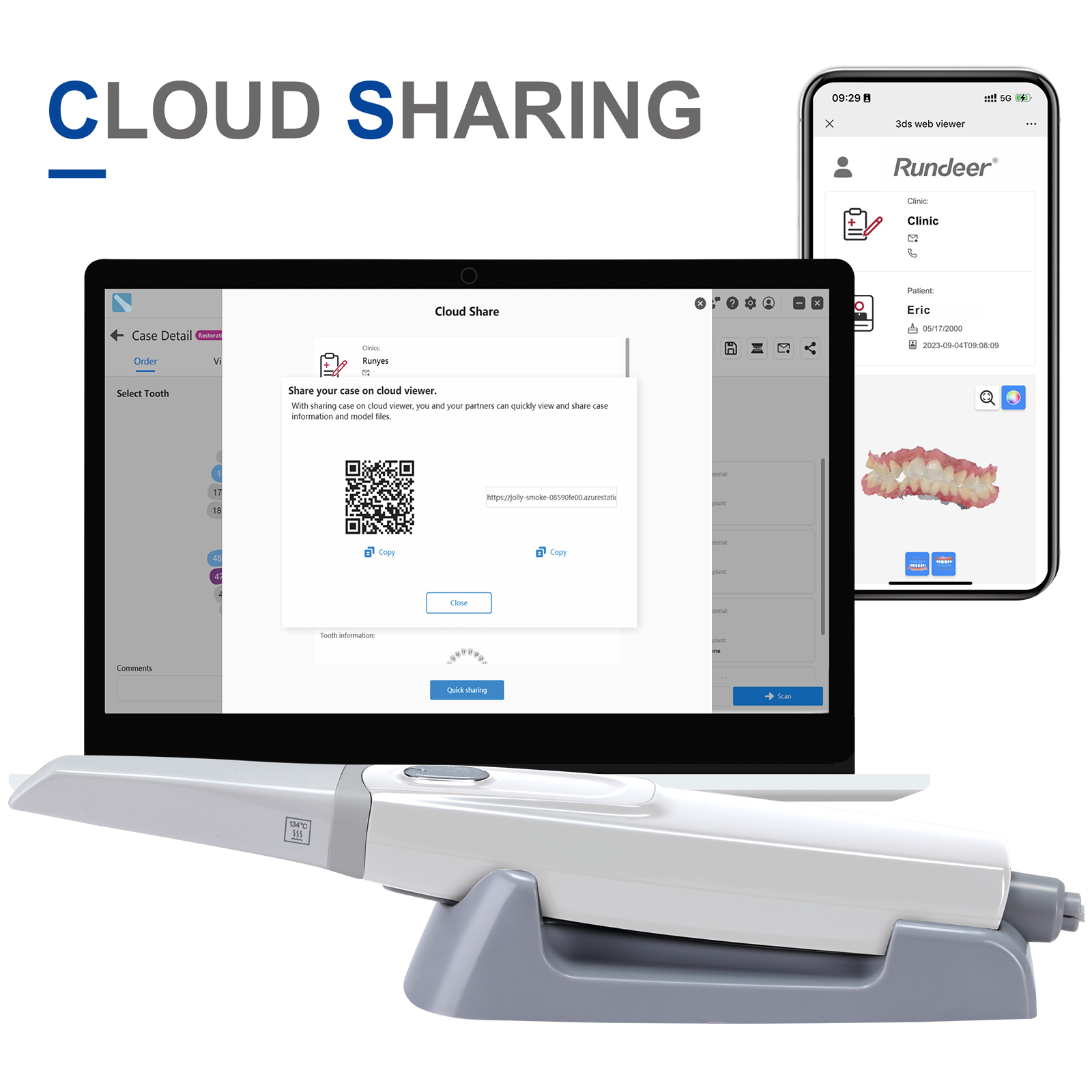
- Case Cloud Sharing
- Orthodontic Simulation Function
FDA Statement:
The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies.
If so, you can bid on this item only if you are an authorized purchaser.
If the item is subject to FDA regulation, I will verify your status as an authorized purchaser of this item before shipping of the item.
FDA for Curing Light: 510(K) Number: K192233, Regulation Number: 21 CFR 872.6070, Product Code: EBZ
FDA for Handpiece: 510(K) Number: K181691, Regulation Number: 872.4200, Classification Product Code: EFB, Subsequent Product Code: EGS
FDA for Scaler: 510(K) Number: K163414, Regulation Number: 872.4850, Product Code: ELC
Feedback:
We commited ourselves to your 100% satisfaction both with our goods and services.
1. Please leave me FIVE star POSITIVE feedback if you are satisfied with our items.
2. Please CONTACT me immediately before leave us neutral feedback or negtive feedback, We will try our best to resolve any problem for you untill you are satisfied.
Notice:
1. Import duties, taxes and charges are not included in the item price or shipping charges. These charges are the buyer’s responsibility.
2. Please check your country’s customs office to determine what these additional costs will be prior to bidding/buying.
Warmly remind:
Pls let me know before taking any action, because your action will has an important impact on our shop.
So pls give me a chance to offer the best sollution for you!!
We commited ourselves to your 100% satisfaction both with our goods and services.
1. Please leave me FIVE star POSITIVE feedback if you are satisfied with our items.
2. Please CONTACT me immediately before leave us neutral feedback or negtive feedback, We will try our best to resolve any problem for you untill you are satisfied.
Notice:
1. Import duties, taxes and charges are not included in the item price or shipping charges. These charges are the buyer’s responsibility.
2. Please check your country’s customs office to determine what these additional costs will be prior to bidding/buying.
Warmly remind:
Pls let me know before taking any action, because your action will has an important impact on our shop.
So pls give me a chance to offer the best sollution for you!!
Our working time:
Monday - Friday 9:00 AM - 5:30PM (Beijing)
Company name: Dreamland Technology Ltd
City, State: Beijing, China
Telephone number: 0371-56645662
Return & Refund:
Return within 30 days is accepted, though the item in good condition.
City, State: Beijing, China
Telephone number: 0371-56645662
Return & Refund:
Return within 30 days is accepted, though the item in good condition.
Copyright © jinlan226