

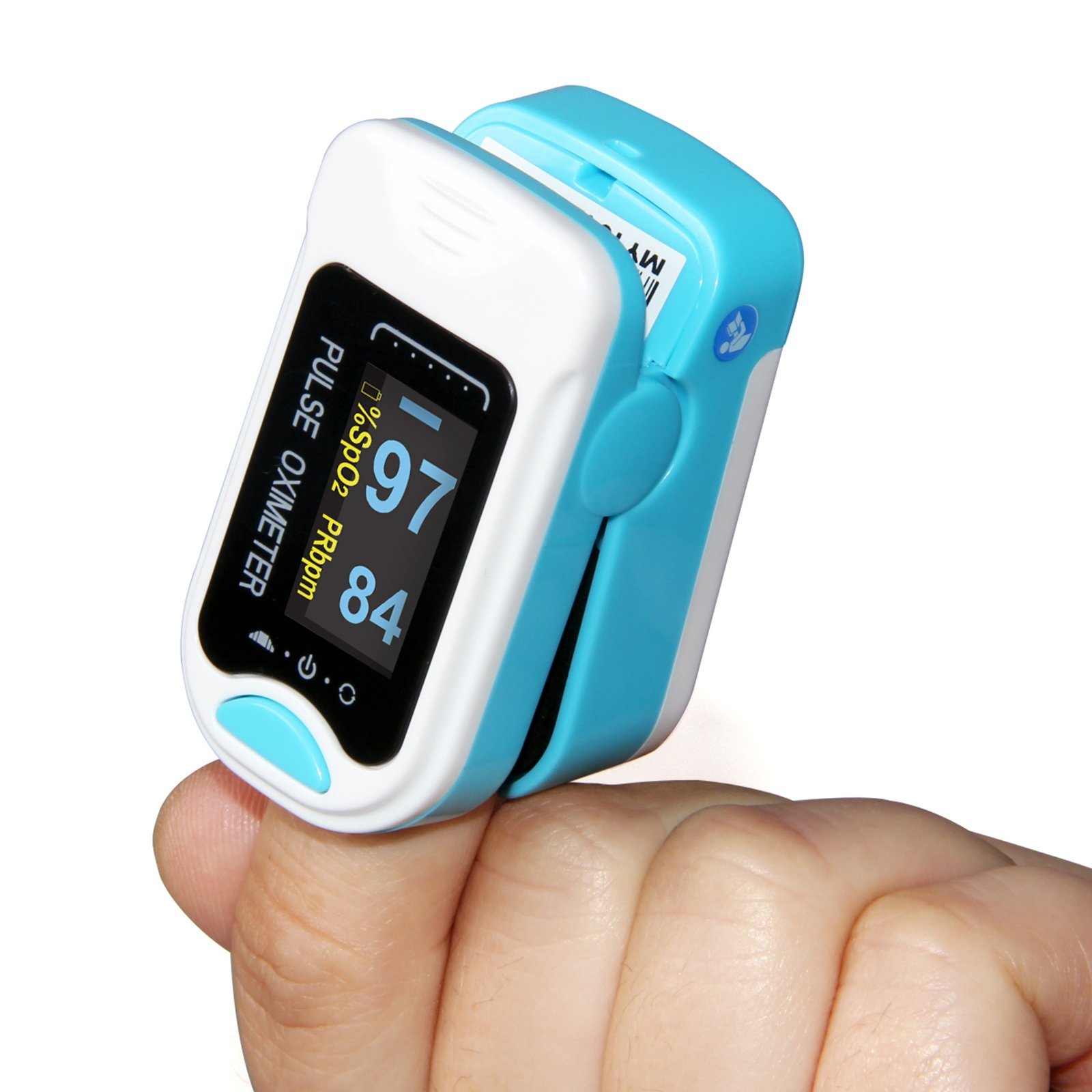

1.Introduction
CMS50NA Pulse
Oximeter is a portable equipment adopting advanced technology, it mainly checks
SpO2 and PR value through the finger, which provides advanced, non-invasive and
scientific means for quantitative measurement of oxygen saturation.
CMS50N
Pulse Oximeter is applicable for use in plateau area, community healthcare,
oxygen bar, Fitness clubs, physical training room, family and medical
institution, etc.
2.Features
1) Can measure SpO2 and Pulse Rate quickly
2) SpO2 and Pulse Rate display,Pulse Rate waveform and bar
graph display
3) Battery voltage low indication
4) The display mode can
be changed.
5) Screen brightness adjustable
3.Performance
SpO2 measurement range:
0%~100%
Accuracy: 70%~100%: ±2%
0%~69%: unspecified
PR measurement
range: 30bpm~250bpm
Accuracy: ±2bpm or ±2%, whichever is
greater.
Resolution:
SpO2: 1%
PR: 1bpm
Measurement Performance in
Weak Filling Condition: SpO2 and pulse rate can be shown correctly when
pulse-filling ratio is 0.4%. SpO2 error is ±4%, pulse rate error is ±2 bpm or
±2%, whichever is greater.
Resistance to surrounding light: The deviation
between the value measured in the condition
of man-made light or indoor
natural light and that of darkroom is less than ±1%.
Working current:
≤30mA
Power supply: two “AAA” batteries
Safety classification: type BF
applied part
4.Physical
characteristic
Dimension: 57mm(L) × 34mm(W) × 31mm(H)
5.Accessory
♦a pulse oximeter
♦a pouch
♦a lanyard
♦a user manual
Buy safe Products
The following FDA Disclaimer is required for all eBay listing in Healthcare category and is included for REFERENCE:
The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies.If the item is subject to FDA regulation,We will verify your status as an authorized purchaser of this item before shipping of the item.If you have questions about legal obligations regarding sales of medical devices, you should consult with the FDA's Center for Devices and Radiological Health.
The Fingertip Pulse Oximeter is registered on the Australian Register of Therapeutic Goods (ARTG) with the code 197923, and certified by FDA of United States and CE,TUV of Europe.The Fingertip Pulse Oximeter that is FDA 510K Approved
Warranty&Return:
1.All items are brand new ,with 1 years warranty.
2.If you have a defective item, you want to return or discount. Please contact us within 2 days from you receive the shipment.We offer 60 days return policy.
3.We will refund the money to you when we get the return items or replace item for you.
4. All return items must be returned with it's original packaging and accessories. Customer is responsible for shipping charges on returned items.
International Buyers - Please Note:
? Import duties, taxes and charges are not included in the item price or shippingcharges. These charges are the buyer's responsibility.
? Please check with your country's customs office to determine what these additional costs will be prior to bidding/buying.

Powered by SoldEazy