Product Feature
⭐This device is used for the fast and precise incision and coagulation of soft tissue and is used in medical institutions.
⭐Safe/Professional/Effective/Convenient.
⭐Incision and coagulation can be adjusted freely.
⭐Seven pieces high performance and hyperfine electrodes.
⭐Reliable and compact unit.
⭐Excellent therapeutic control.
⭐The whole set of electrode holder is light and autoclavable.
⭐Innovative conductive bracelet (Neutral Electrode).
⭐30 watts is enough for greater efficiency.
Product Specification
Model: Electrosurgery Unit ES-20
Supply voltage: 115VAC / 230VAC
Supply frequency: 50 Hz / 60Hz
Power consumption: 170 VA ~ 230 VA
Max power output: 30W
Characteristic impedance: 600Ω
Output impedance range: 100Ω ~ 2000Ω
Output voltage: 650V PP - P = 10, W=1
Output frequency: 1.2MHz ± 0.2MHz
Main unit dimensions: 23 x 26 x 11cm
Main unit weight: 1.5kg
Net weight: 2.0kg
Gross weight: 3.0kg
Box dimensions: 37 x 37 x 17cm
Operating temperature: +10℃ to +30℃
Storage temperature: -20℃ to +70℃
Operating relative humidity: 30% to 75%
Storage relative humidity: 10% to 100% including condensation
Atmospheric pressure: 80KPa to 106KPa
Altitude: ≤2000 meters
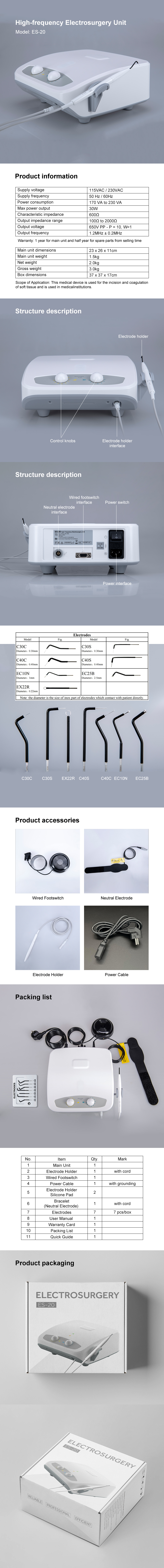
Package Included
1 x Main Unit
1 x Electrode Holder(with cord)
1 x Wired Footswitch
1 x Power Cable(with grounding)
2 x Electrode Holder Silicone Pad
1 x Bracelet (Neutral Electrode)
7 x Electrodes
1 x User Manual
1 x Warranty Card
1 x Packing List
1 x Quick Guide
FDA Statement :
The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies.
If so, you can bid on this item only if you are an authorized purchaser.
If the item is subject to FDA regulation, I will verify your status as an authorized purchaser of this item before shipping of the item.
510(K) Number: K192233
Regulation Number: 21 CFR 872.6070
Product Code: EBZ
FDA for Handpiece:
510(K) Number: K181691
Regulation Number: 872.4200
Classification Product Code: EFB
Subsequent Product Code: EGS
FDA for Scaler:
510(K) Number: K163414
Regulation Number: 872.4850
Product Code: ELC
FDA for Handpiece:
510(K) Number: K181691
Regulation Number: 872.4200
Classification Product Code: EFB
Subsequent Product Code: EGS