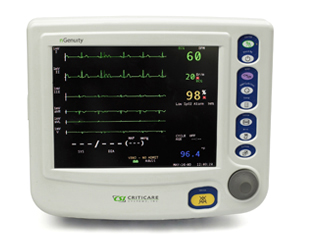
Optional features: End-tidal and inspired CO2, arrhythmia and ST analysis and integrated printer.
Optional CO2 monitoring incorporates CSI’s patented water trap design with full occlusion clearing.
Mini-Trends display of condensed tabular vital signs data to show recent monitoring activity.
Compatible with popular Electronic Medical Records (EMR) systems for both large and small offices.
Easy to read 10.4 inch TFT display.
Easy to learn, easy to use interface with simplified menus and dedicated function key
DISCLAIMER:
NOTE:
CE MARKED - This product is in compliance with the essential requirements of Council Directive 93/42/EEC Medical Device Directive, as amended by Council Directive 2007/47/EC, class IIa
NOTE: The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. If so, do not bid on this item unless you are an authorized purchaser. If the item is subject to FDA regulation, We will verify your status as an authorized purchaser of this item before shipping of the item.